Osmo and The Science of Hydration
Pioneers of A Revolution in Sports Nutrition
Why is Osmo more effective than competing sports drinks? Because Science!
It's all about fine-tuning the types of sugar and salt, as well as their ratios. First, a quick review on how your body absorbs water.
When two areas are segregated by such a semi-permeable membrane (i.e. intestinal tissue), they want to achieve equilibrium. So what happens in your gut? Water solutions travel from areas of low concentration to areas of high concentration to even things out. The "osmolality" of a solution is the percentage of "stuff" in said solution. (For our purposes, Osmolality is the same as Specific Gravity - for you beer/chemistry enthusiasts)
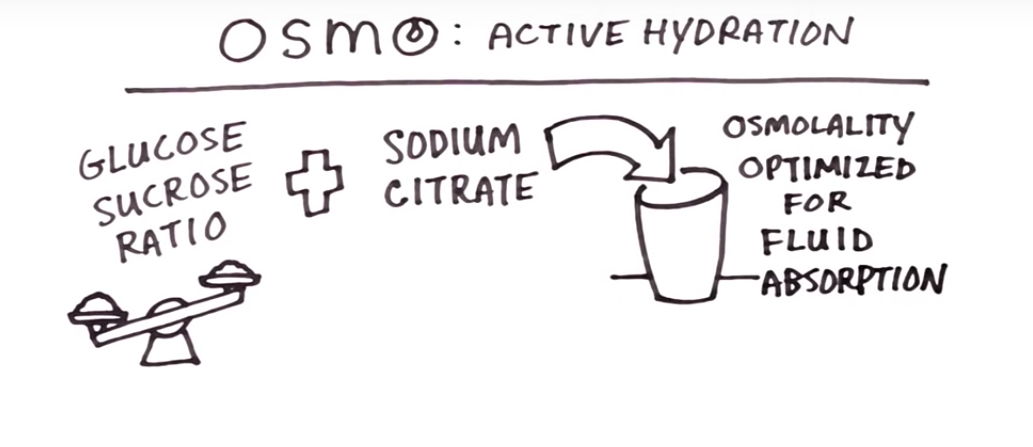
In a sports drink, the osmolality of a solution is primarily determined by the amount of carbohydrates, amino acids, and proteins. Electrolytes play a secondary role. In the bloodstream, osmolality is determined by substances like sodium, potassium, amino acids, and glucose. The idea of Osmo Active Hydration is to optimize intestinal fluid exchange for more effective hydration. Doing this is as simple as making the concentration of the drink lower than that of the blood; precisely what Osmo has done.
Osmo's effective differentiator is the 3.2% solution, or 3.2 grams of carbohydrates per 100ml solution. To maximize efficiency for the small amount of solids in solution, Osmo Active Hydration uses a finely-tuned ratio of glucose to sucrose, and uses sodium citrate rather than the usual sodium chloride, which is easier on most people's stomachs.
For other drinks with higher osmolality, your intestines use extra body water to to dilute the solution, so it can then be re-absorbed. This is a slower process, which can result in a "bloated" feeling and reduces the overall amount of fluid that can be absorbed, thereby reducing hydration.
Hydration is performance, whether you're challenging yourself of contesting a course record. When you're working hard, you're losing water; and there's no more efficient way to replace it than with Osmo Active Hydration.